 They were oxygen, hydrogen, nitrogen, carbon monoxide, methane, and nitric oxide. Six gases, however, resisted every attempt at liquefaction and were thus known at the time as permanent gases. His procedure consisted of cooling the gas by immersion in a bath of ether and dry ice and then pressurizing the gas until it liquefied. One of these scientists, English physicist and chemist Michael Faraday (1791 –1867), had succeeded, by 1845, in liquefying most of the gases then known to exist. The development of cryogenics as a low temperature science is a direct result of attempts by nineteenth century scientists to liquefy the permanent gases. 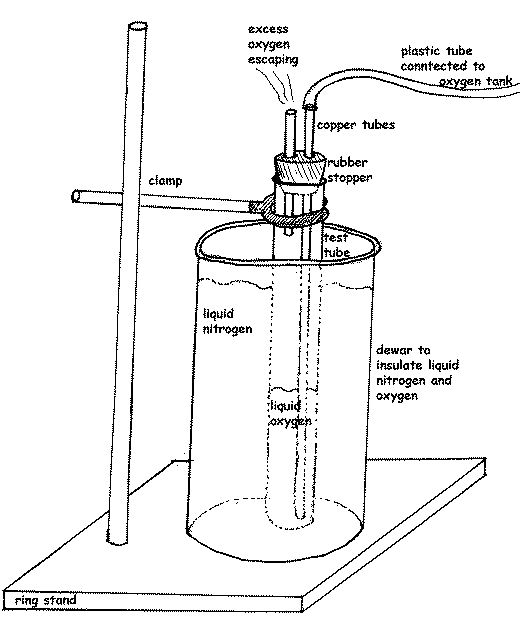
Of superfluidity, in which it can flow through exceedingly narrow passages with no friction. Very near absolute zero (2.2K) liquid helium undergoes a transition to a state Table 1. In addition, many metals and ceramics lose all resistance to the flow of electricity, a phenomenon called superconductivity. For example, rubber, most plastics, and some metals become exceedingly brittle, and nearly all materials contract. 
The mechanical and electrical properties of many materials change very dramatically when cooled to 100K or lower. In addition to studying methods for producing and maintaining cold environments, the field of cryogenics has also come to include studying the properties of materials at cryogenic temperatures. Cryogenics, then, deals with producing and maintaining environments at temperatures below about 173K ( –148 ☏). Thus, –459 ☏ corresponds to –273 ☌ corresponds to 0K (note that by convention the degree symbol is omitted, so that 0K is read zero Kelvin). It is the basis of an absolute temperature scale, called the Kelvin scale, whose unit, called a Kelvin (K) rather than a degree, is the same size as the Celsius degree. The existence of absolute zero was first pointed out in 1848 by British mathematician and physicist William Thomson, Lord Kelvin (1824 –1907), and is now known to be –459 ☏ ( –273 ☌). Absolute zero represents the lowest attainable temperature and is associated with the complete absence of atomic and molecular motion. The temperature of a sample, whether it be a gas, liquid, or solid, is a measure of the energy it contains, energy that is present in the form of vibrating atoms and moving molecules. (Absolute zero is defined as the lowest temperature possible, a state where no heat energy exists.) Consequently, the term cryogenic applies to temperatures from approximately –148 ☏( –100 ☌) down to absolute zero. The origin of cryogenics as a scientific discipline coincided with the discovery by nineteenth century scientists, that the permanent gases can be liquefied at exceedingly low temperatures. Among others, they include oxygen (O), nitrogen (N), hydrogen (H), and helium (He). More specifically, a low-temperature environment is termed a cryogenic environment when the temperature range is below the point at which permanent gases begin to liquefy. The word cryogenics comes from the Greek word kryos, meaning ”cold “ combined with a shortened form of the English verb ”to generate, “it has come to mean the generation of temperatures well below those of normal human experience. 
Laser cooling and Bose-Einstein condensateĬryogenics is the science of producing and studying low-temperature environments. Methods of producing cryogenic temperatures 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
